A Tour of the Anhui Ma’anshan Manufacturing Site
FDA/EMA/NMPA-compliant, high-containment CDMO services — from early-phase process development through commercial-scale GMP manufacturing of the world’s most complex & potent APIs and ADC payload-linkers.
- OEB 5 Containment Level
- 0.01 μg/m³ OEL Capability
- 4,000L+ HPAPI GMP Capacity
- 5 Dedicated HPAPI Lines
A Purpose-Built HPAPI & ADC CDMO Site
Located in Anhui Province, China, the Ma’anshan GMP manufacturing complex is a purpose-designed facility for high-potency API synthesis, ADC payload-linker production and GMP-grade pharmaceutical intermediates. Spanning approximately 142,000 ft², it combines world-class containment engineering with the flexibility to serve clients from kilogram to metric tonne commercial supply.
Two complementary sites operate in tandem: Site 1 focuses on HPAPI, ADC payloads, and contained process chemistry; Site 2 handles peptide process development, APIs, GMP intermediates and RSMs at larger scale: together creating a fully integrated CDMO solution under a single quality system.

Regulatory compliance spans FDA, EMA and NMPA GMP standards, supported by successfully completed European QP qualification audits (2023) with a pipeline of PMDA and additional inspections scheduled through 2026.
HPAPI Synthesis
5 dedicated OEB 5 GMP production lines with a combined reactor volume exceeding 4,000L. Negative pressure isolators for sub-nanogram OEL work (0.01 μg/m³).
ADC Payload-Linker Manufacturing
GMP synthesis of vcMMAE, Exatecan Mesylate, Trabectedin and related ADC building blocks — with FDA DMF filings across the full series.
API & Intermediate Scale-Up
~175 reactors (100L–8,000L) with 480,000L total volume at Site 2. Commercial scale kilograms to tonnes for APIs, GMP intermediates and RSMs.
Purification & Finishing
Multiple preparative HPLC systems and lyophilisers, supporting isolation of complex natural product analogues, peptide payloads, and sensitive APIs.
Reaction Capabilties
Wide range of reaction conditions and techniques: hydrogenations, oxidations, air & moisture sensitive chemistry and photochemical reactions alongside extreme temperature ranges (−80 °C to +240 °C) utilising state-of-the-art equipment.
Quality & Compliance
FDA/EMA/NMPA GMP. EU QP audit passed 2023. Japan PMDA inspection scheduled 2026. Multiple drug manufacturing licences. >60 successful client audits to date.
Both sites are engineered for operational efficiency, safety and regulatory robustness. Key design features include:
Containment & Ventilation
OEB 3/4/5 design. All-supply/all-return fresh air in synthesis areas. Local Exhaust Ventilation (LEV). HEPA-filtered clean rooms (Class D).
Process Control
DCS (Distributed Control System) for full process monitoring. Gravity-flow 4-floor design maximises operational efficiency.
Utilities
10kV dual power supply. Glycol refrigeration system. Liquid nitrogen + generator. Purified water (USP/JP compliant, 3 m³/h). Steam at 0.8 MPa / 160 °C.
Supporting Equipment
Centrifuges, including peeler and inverting filter types; drying equipment, (double-cone, vacuum trays, Filter-Wash-Dryer); downstream processing, including jet- and hammer mills.
Waste Treatment
600 MT/day wastewater treatment plant (WWTP). Regenerative Thermal Oxidisation (RTO) for non-halogen waste gas; resin adsorption for halogenated streams. Compliant environmental management.
Materials of Construction
Reactors available in Glass-Lined and Stainless Steel, covering a wide range of chemical compatibility requirements.
Phase II Expansion
~27,000 m² expansion planned for additional ADC Payload-Linker and HPAPI capacity, with ~51,000L additional reactor volume.

Regulatory Track Record
2022
Site commences GMP operations. Initial client audit programme begins.
2023
European regulatory qualification inspeciton; over 50 client audits and more than 20 client visits.
2024
2 drug manufacturing license inspections; more than 60 client audits and over 30 client visits.
2025
Two additional drug manufacturing licence inspections; GMP Inspection; more than 40 client audits and over 30 client visits.
2026
Onsite Japan PMDA inspection scheduled
ADC Payloads & HPAPI Series — GMP Supply with FDA DMF
The following highly potent APIs and ADC payload-linker series are available for GMP supply. Many carry active FDA Drug Master File (DMF) or ASMF filings, enabling direct regulatory referencing for client submissions.
vcMMAE Series (Auristatin ADC Payload)
Key ADC cytotoxic payload for oncology. Full series from MMAE building blocks to finished MC-Val-Cit-PAB-MMAE.
| CAS No. | Compound Name | Structure | FDA DMF No. |
|---|---|---|---|
| 474645-27-7 | MMAE | 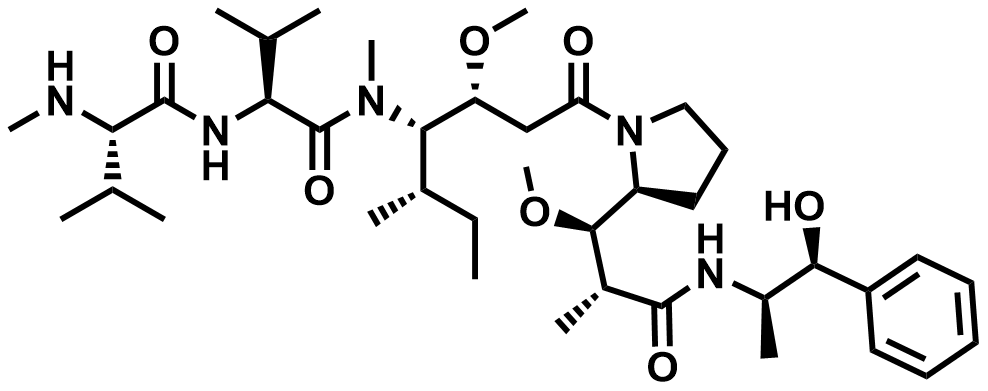 | MF# 036741 |
| 646502-53-6 | MC-Val-Cit-PAB-MMAE |  | MF# 036740 |
| 159857-80-4 | MC-Val-Cit-PAB (Intermediate) | 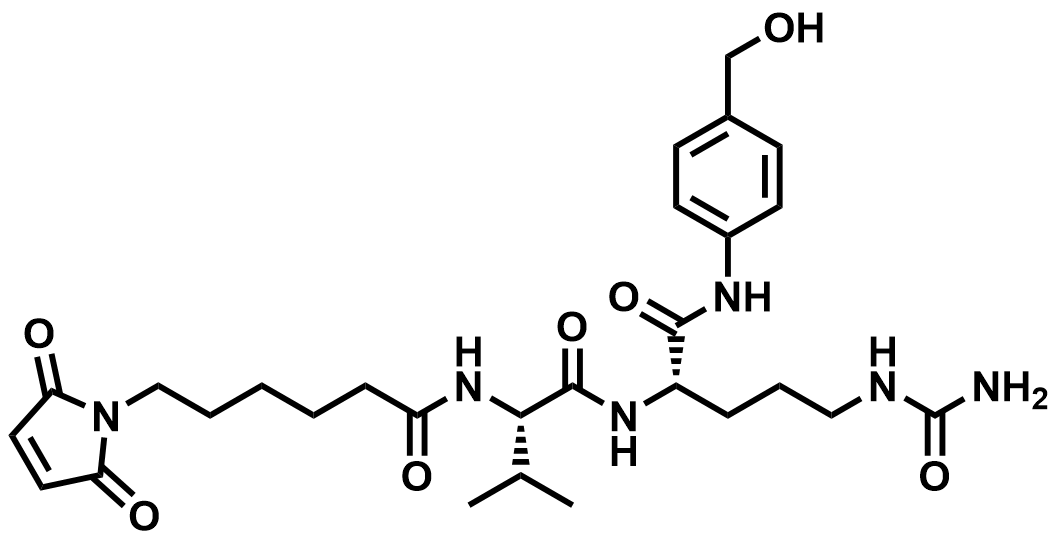 | MF# 035550 |
| 160800-65-7 | Boc-Dap-NE (MMAE Intermediate) | 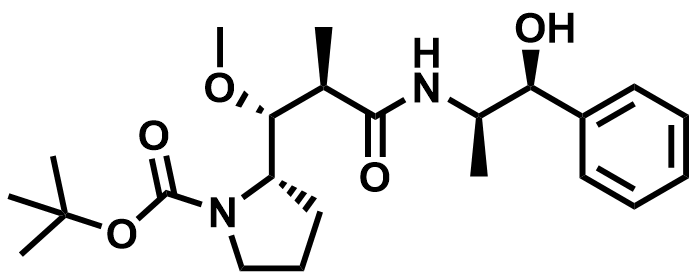 | MF# 035548 |
| 863971-44-2 | Fmoc-3VVD-OH (MMAE Intermediate) | 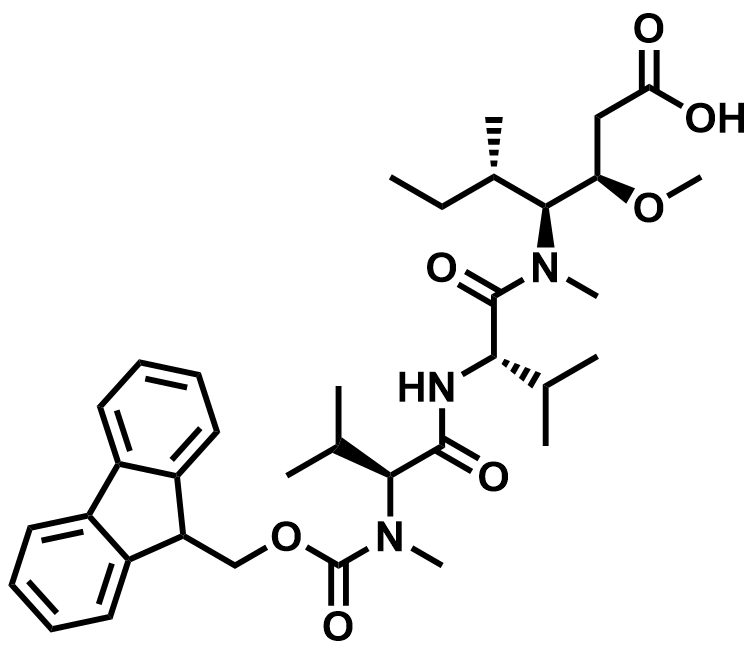 | MF# 035549 |
Exatecan Series (Camptothecin ADC Payload)
Topoisomerase I inhibitor payload increasingly used in next-generation ADCs.
| CAS No. | Compound Name | Structure | FDA DMF No. |
|---|---|---|---|
| 169869-90-3 | Exatecan Mesylate | 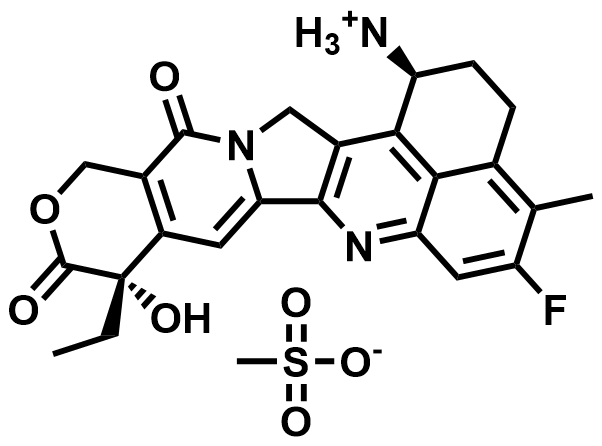 | MF# 036708 |
| 182182-31-6 | Exatecan Intermediate 01 | 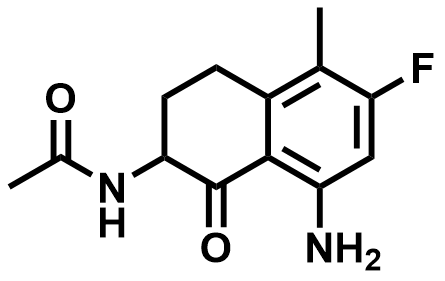 | MF# 037931 |
| 110351-94-5 | Exatecan Intermediate 02 | 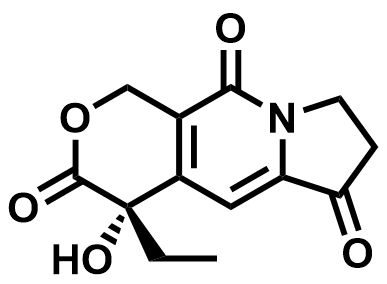 | MF# 037930 |
Trabectedin Series (Ecteinascidin-743) — Soft Tissue Sarcoma / Ovarian Cancer
Complex marine-derived natural product. EMA-approved indication. US DMF and ASMF filed.
| CAS No. | Compound Name | Structure | DMF Reference |
|---|---|---|---|
| 114899-77-3 | Trabectedin | 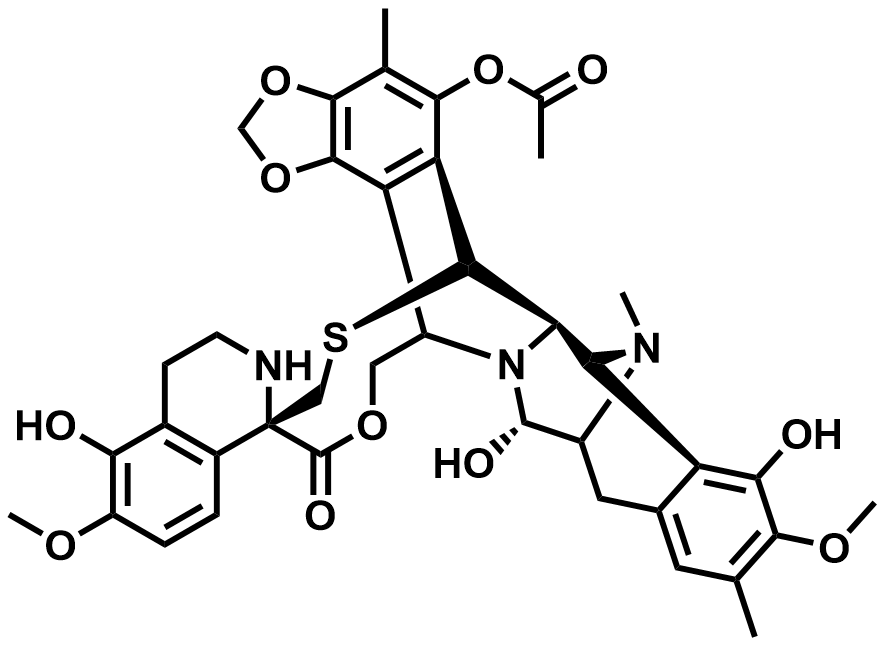 | US DMF 036724 / ASMF 00518 |
| 182201-71-4 | Trabectedin Key Intermediate | 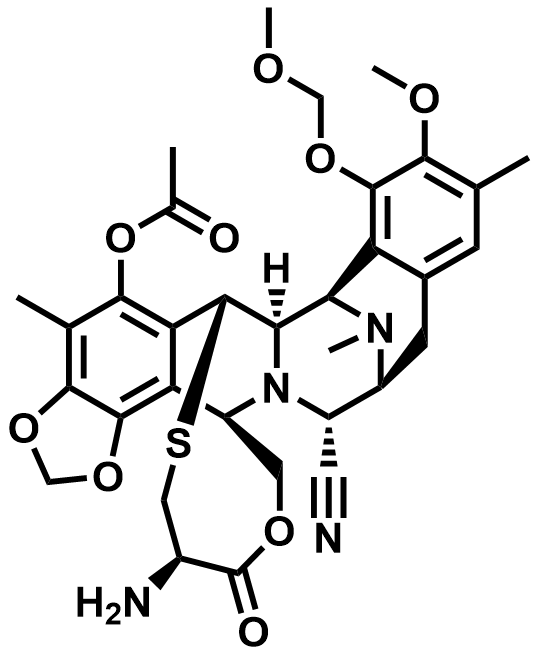 | MF# 036899 |
Eribulin Series — Breast Cancer HPAPI
Highly complex macrocyclic HPAPI. US DMF and ASMF filed. Full intermediate series available.
| AS No. | Role | Structure | DMF Reference |
|---|---|---|---|
| 441045-17-6 | Eribulin Mesylate (API) | 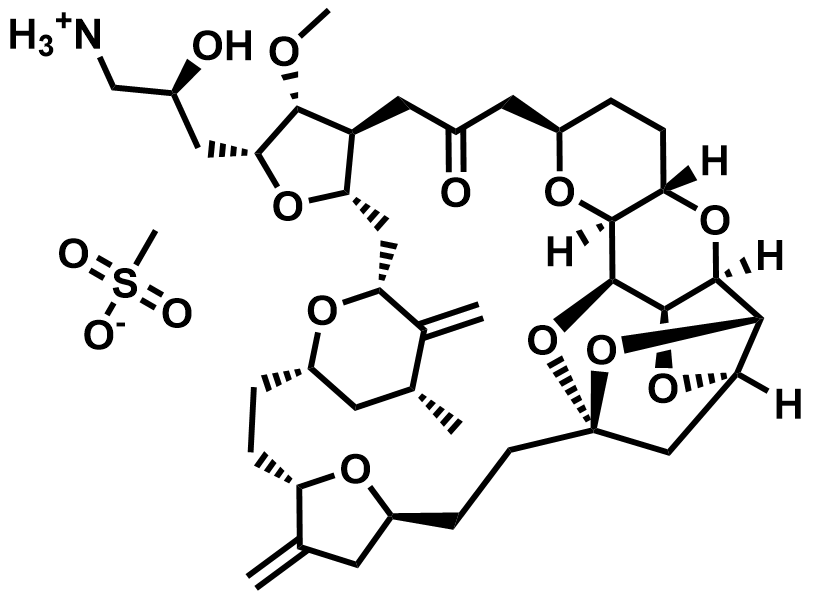 | US DMF 037559 / ASMF 04340 |
| 157322-83-3 | Eribulin Intermediate | 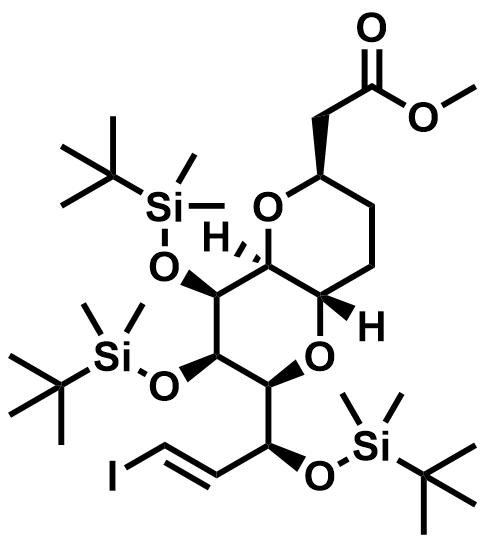 | DMF# 037054 |
| 2835443-65-5 | Eribulin Intermediate | 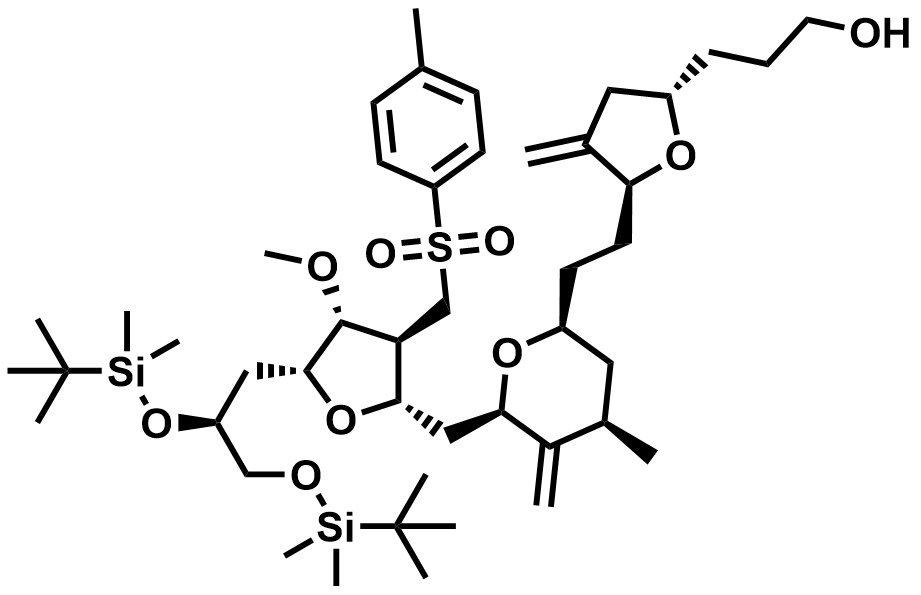 | DMF# 033836 |
| 1960458-56-3 | Eribulin Intermediate | 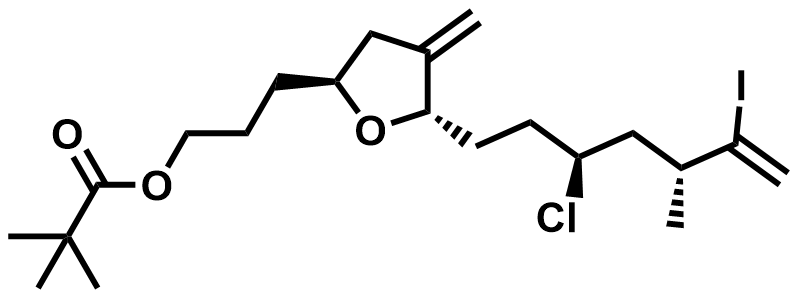 | Commercial |
| 157322-47-9 | Eribulin Intermediate | 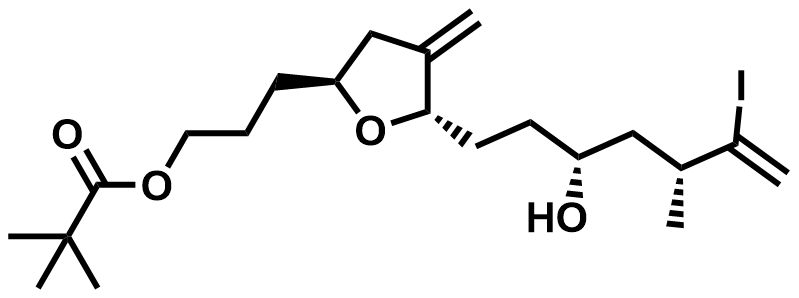 | Commercial |
| 1808153-91-4 | Eribulin Intermediate | 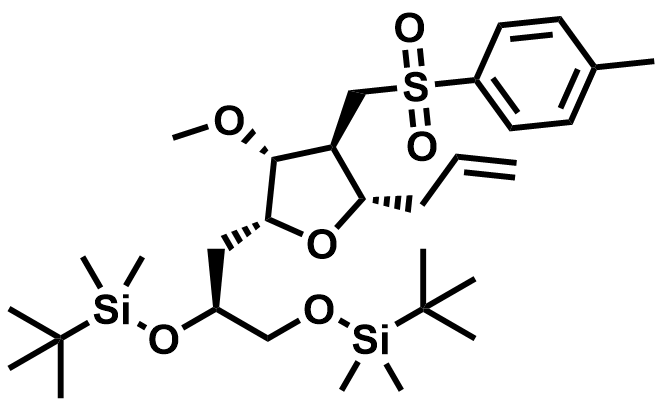 | Commercial |
| 1808153-90-3 | Eribulin Intermediate | 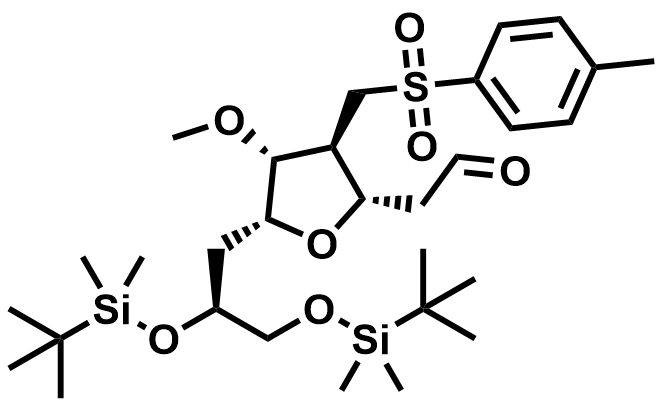 | Commercial |
| 253128-15-3 | EribulinN-3 | 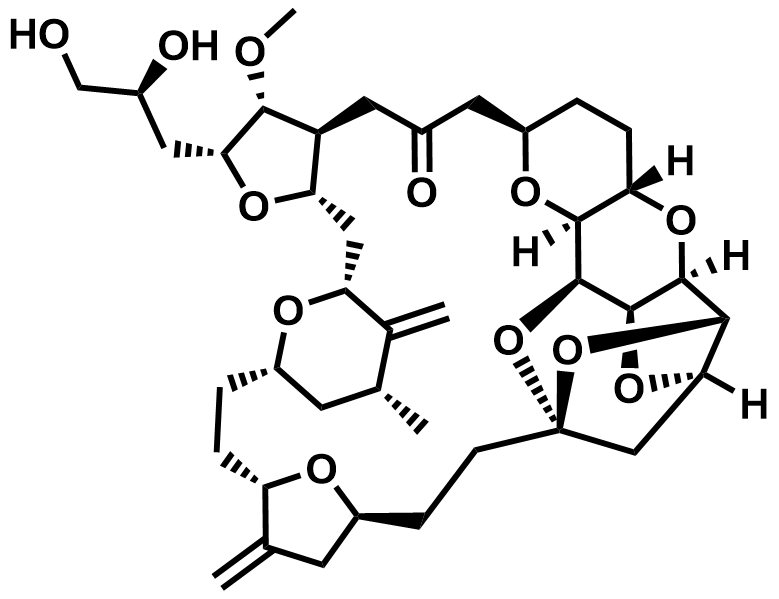 | Commercial |
Oncology APIs & GMP Intermediates
| CAS No. | Product | Structure | Indication | Status |
|---|---|---|---|---|
| 571190-30-2 | Palbociclib | 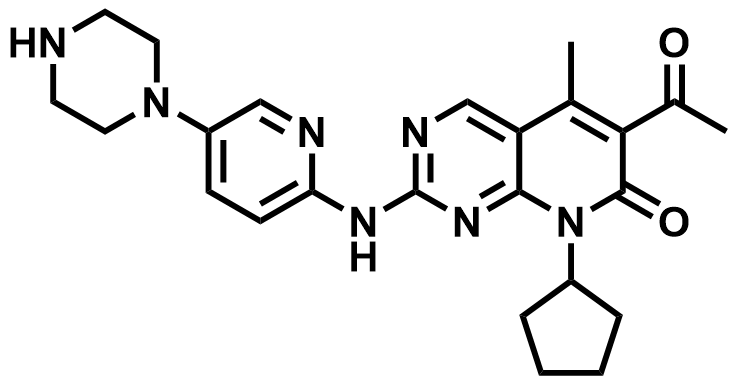 | Breast cancer (CDK4/6i) | DMF in Preparation |
| 1211441-98-3 | Ribociclib | 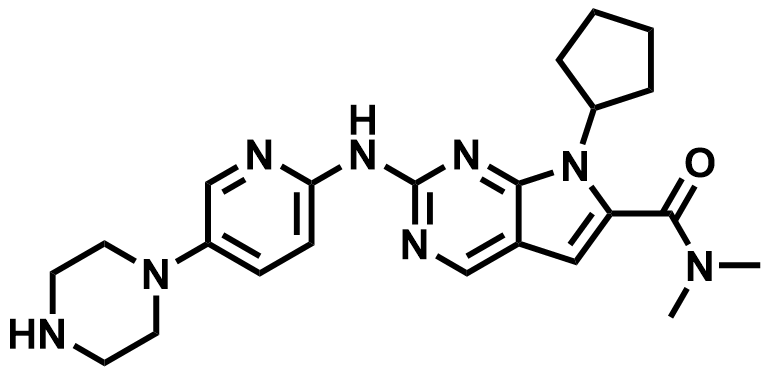 | Breast cancer (CDK4/6i) | Filing Q4 2026 (CN/US/EU) |
| 497871-47-3 | Lurbinectedin | 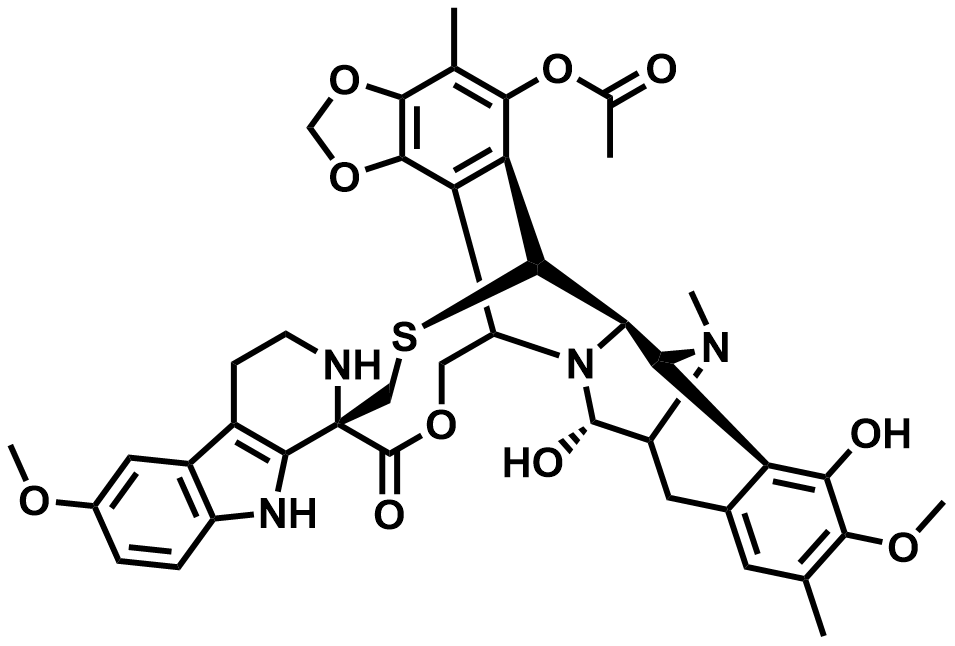 | Non-small cell lung carcinoma | DMF in Preparation |
| 1013916-37-4 | 2-chloro-8-cyclopentyl-5-Methylpyrido[2,3-d]pyrimidin-7(8H)-one | 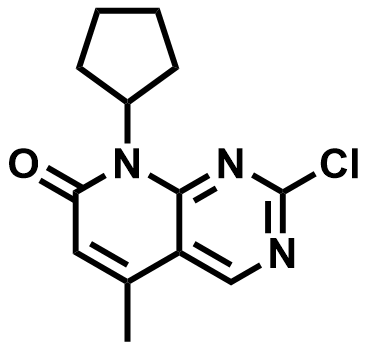 | Palbociclib Intermediate | Commercial |
| 1016636-76-2 | 6-bromo-2-chloro-8-cyclopentyl-5-Methylpyrido[2,3-d]pyrimidin-7(8H)-one | 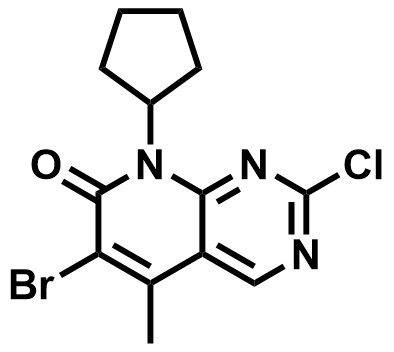 | Palbociclib Intermediate | Commercial |
| 571188-59-5 | tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate | 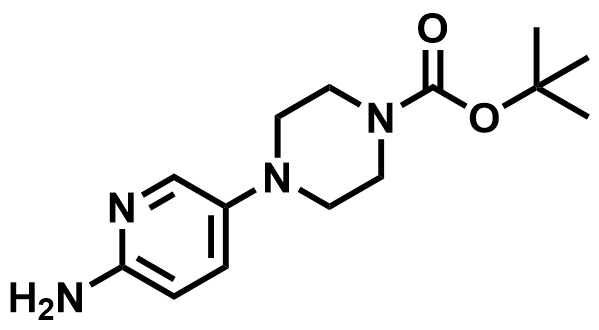 | Palbociclib Intermediate | Commercial |
| 571189-16-7 | 1-BOC-4-(6-Nitropyridin-3-yl)piperazine | 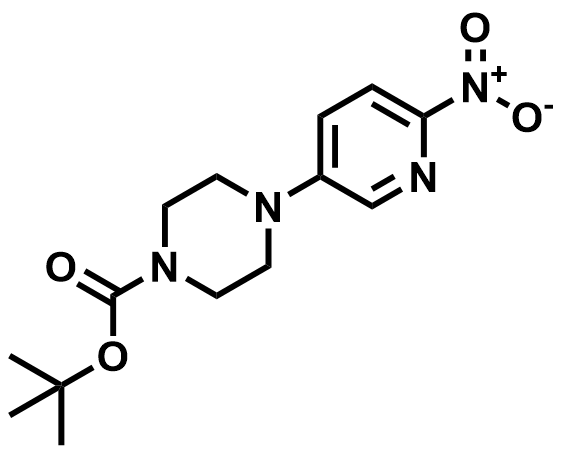 | Palbociclib Intermediate | Commercial |
Vitamin D Analogues & Musculoskeletal HPAPIs
A broad portfolio of Vitamin D analogue APIs and their GMP intermediates — covering calcitriol, calcipotriol, alfacalcidol, paricalcitol, eldecalcitol, maxacalcitol, doxercalciferol, tacalcitol and calcifediol. Multiple DMF filings including a PMDA-registered DMF for Bazedoxifene Acetate.
| CAS No. | Product | Structure | Therapy Area | Status |
|---|---|---|---|---|
| 112849-27-1 | Calcipotriol Intermediate (G10) | 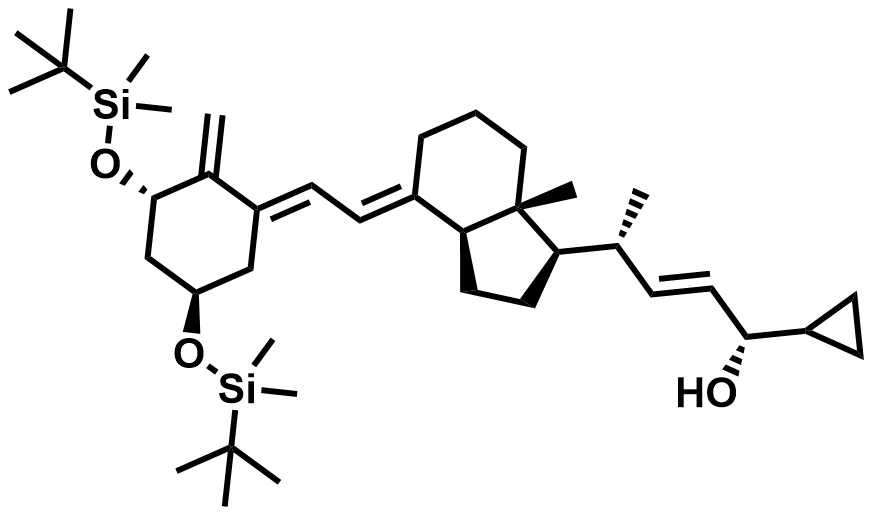 | Psoriasis | DMF# 029298 / 038367 |
| 112965-21-6 | Calcipotriol | 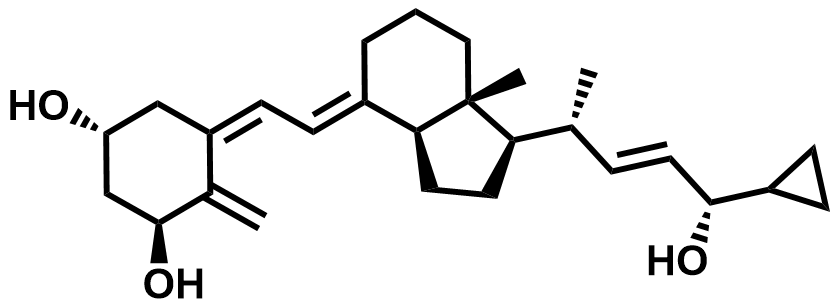 | Psoriasis | DMF in Preparation |
| 113082-99-8 | Calcipotriol Impurity C | 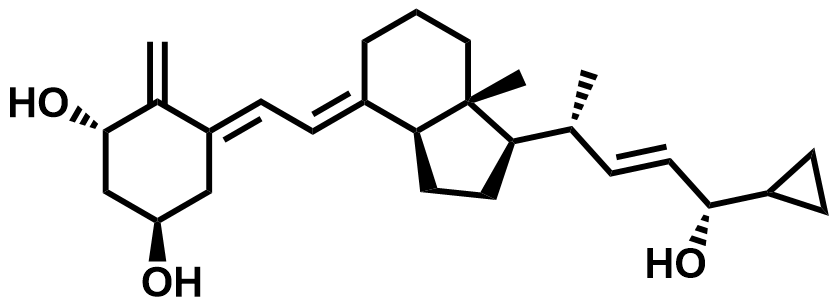 | Intermediate | Commercial |
| 41294-56-8 | Alfacalcidol | 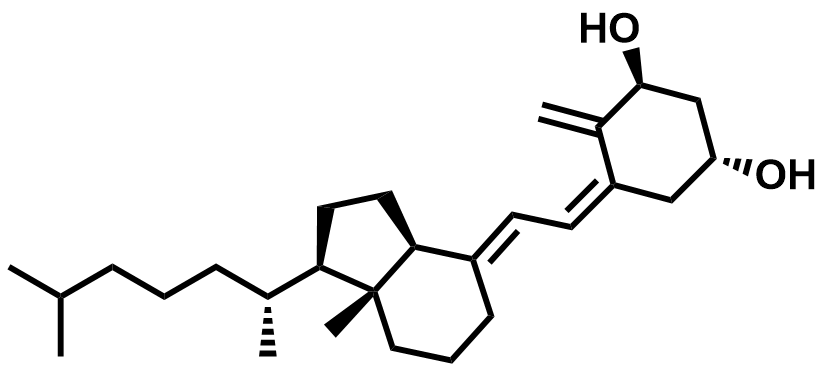 | Osteoporosis / Rickets | Pilot |
| 112670-85-6 | Alfacalcidol Intermediate | 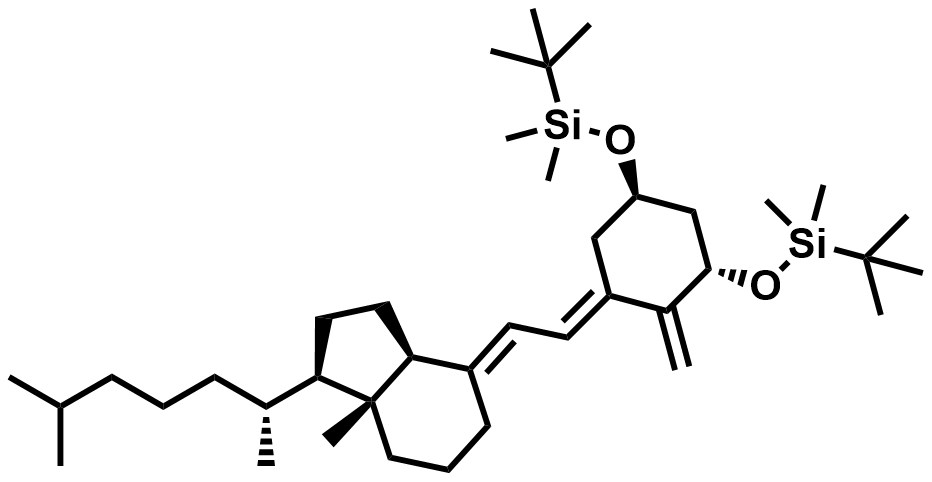 | Intermediate | Commercial |
| 32222-06-3 | Calcitriol | 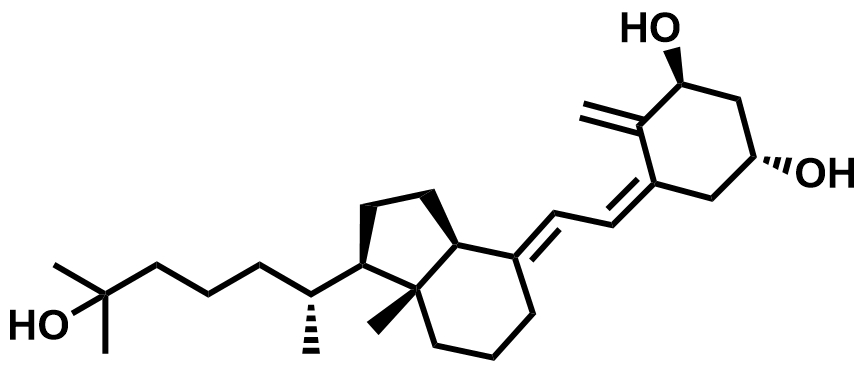 | Osteoporosis / Renal | Pilot |
| 128387-35-9 / 73837-24-8 / 140710-96-9 | Calcitriol Intermediates | 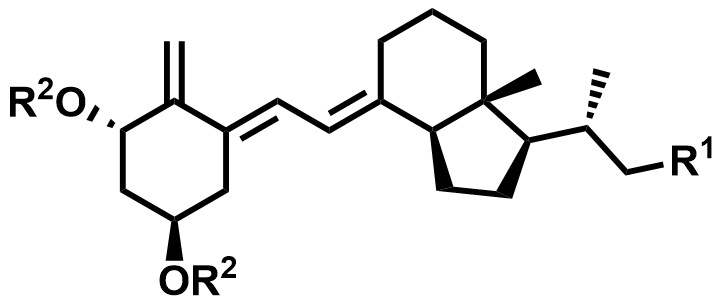 | Intermediates | Commercial |
| 63283-36-3 | Calcifediol Monohydrate | 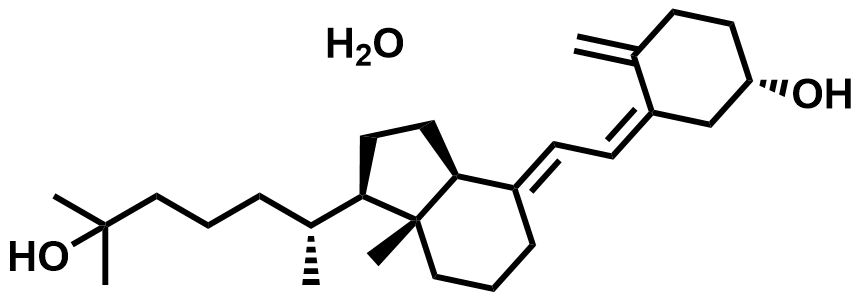 | Rickets / Hypoparathyroidism | Pilot |
| 104121-92-8 | Eldecalcitol | 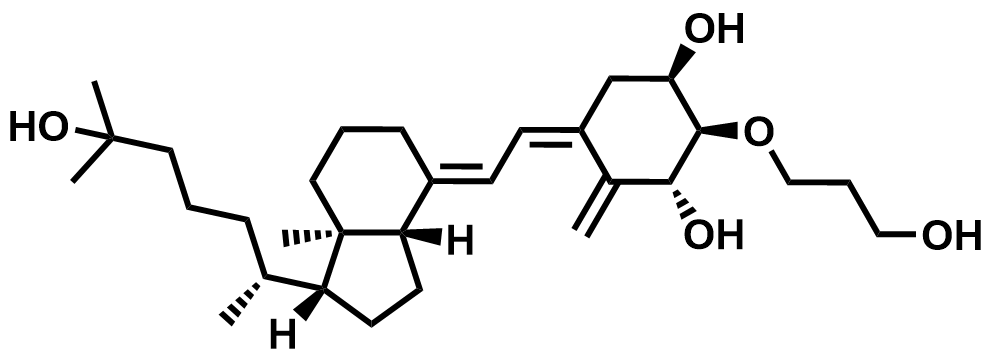 | Osteoporosis | Pilot |
| 200636-42-6 / 2189732-17-8 / 200636-54-0 | Eldecalcitol Intermediates | 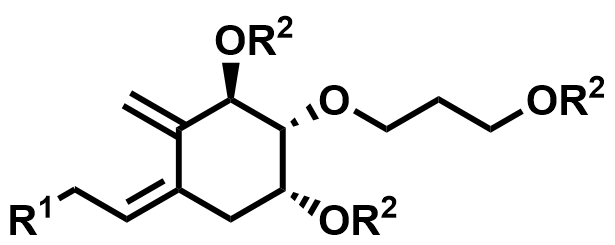 | Intermediates | Commercial |
| 66774-84-3 / 70550-73-1 / 144848-24-8 | Eldecalcitol Intermediates | Intermediates | Commercial | |
| 81506-24-3 / 81522-68-1 | Maxacalcitol Intermediates | 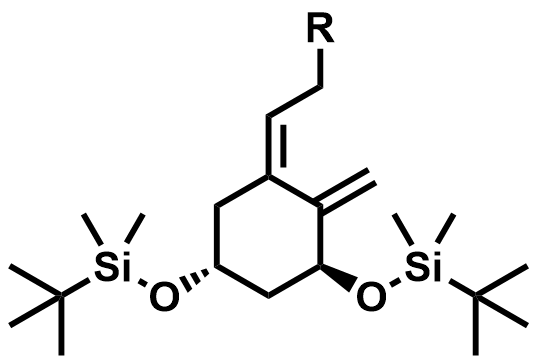 | Intermediates | Commercial |
| 884488-07-7 / 192573-33-4 | Maxacalcitol Intermediates | 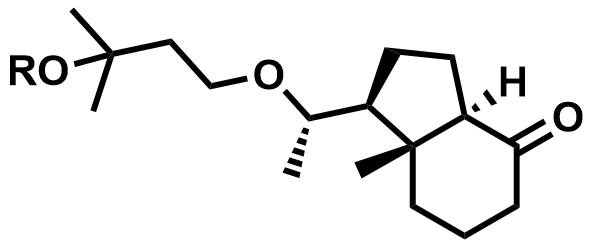 | Intermediates | Commercial |
| 54573-75-0 | Doxercalciferol | 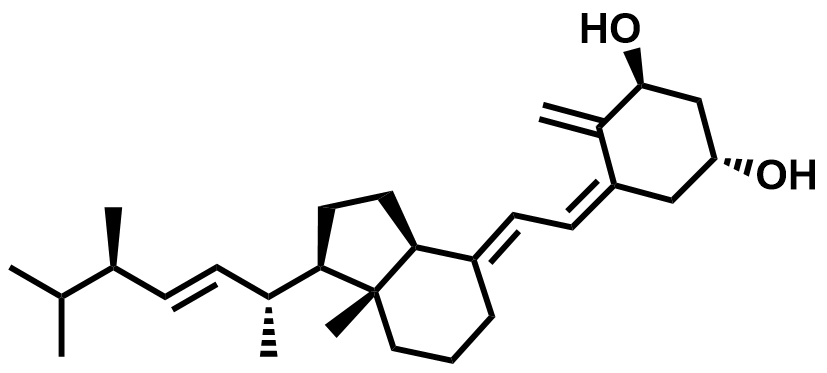 | Secondary hyperparathyroidism | Pilot |
| 131918-61-1 | Paricalcitol | 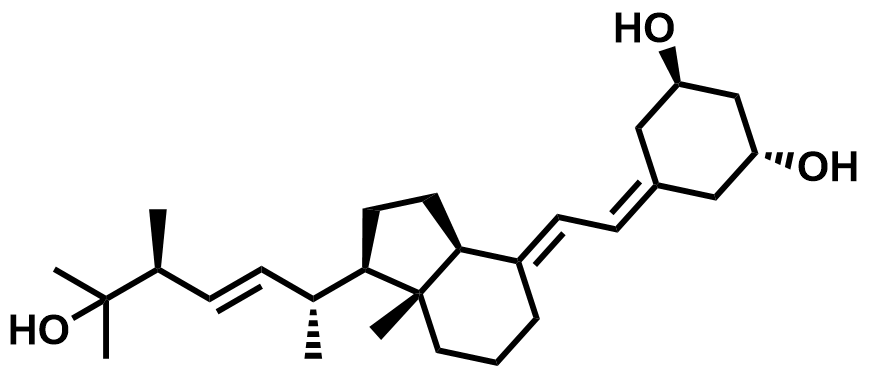 | Secondary hyperparathyroidism | Pilot |
| 57333-96-7 | Tacalcitol | 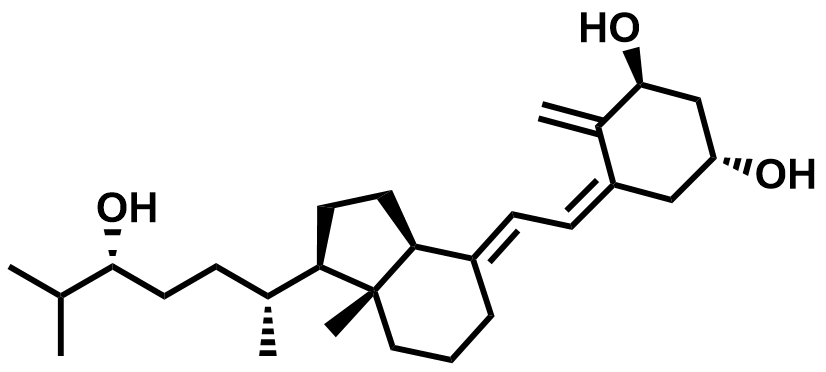 | Psoriasis | Pilot |
| 198481-33-3 | Bazedoxifene Acetate | 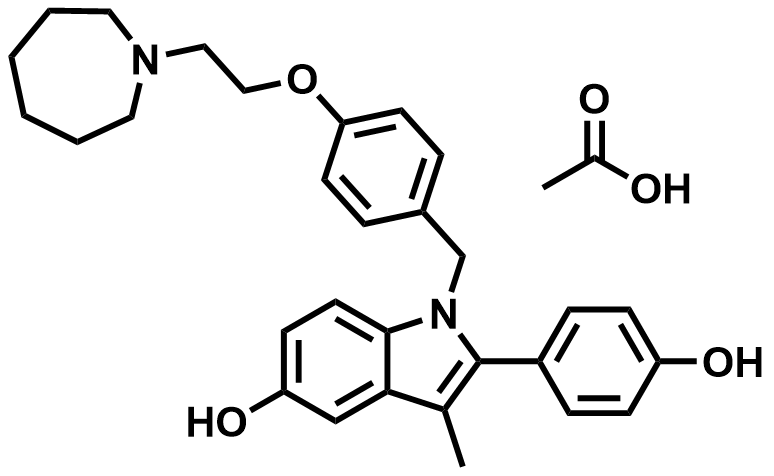 | Postmenopausal osteoporosis | Japan DMF 302MF10051 |
Cardiovascular, CNS, Gastrointestinal & Anti-Infective APIs
| CAS No. | Product | Structure | Indication | Status |
|---|---|---|---|---|
| 274693-27-5 | Ticagrelor | 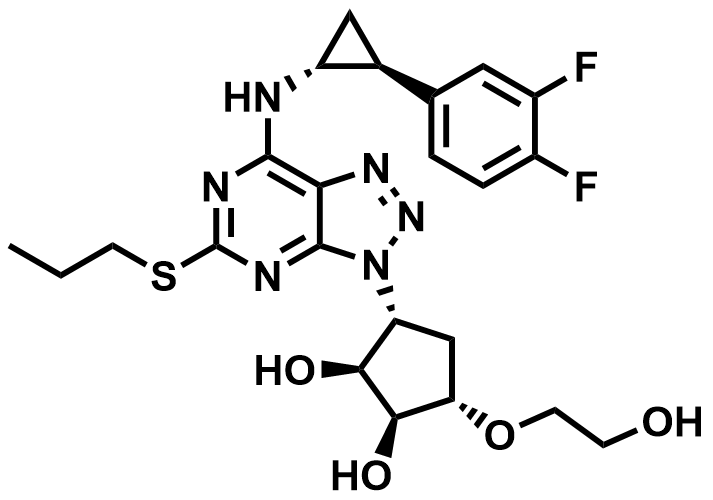 | Acute coronary syndromes | Pilot |
| 145783-15-9 | Ticagrelor Intermediate | 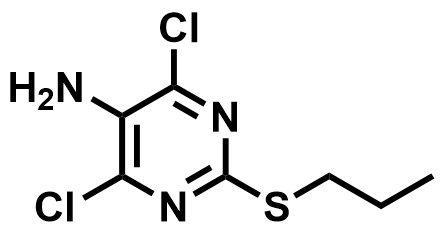 | Intermediate | Commercial |
| 376608-65-0 | Ticagrelor Intermediate | 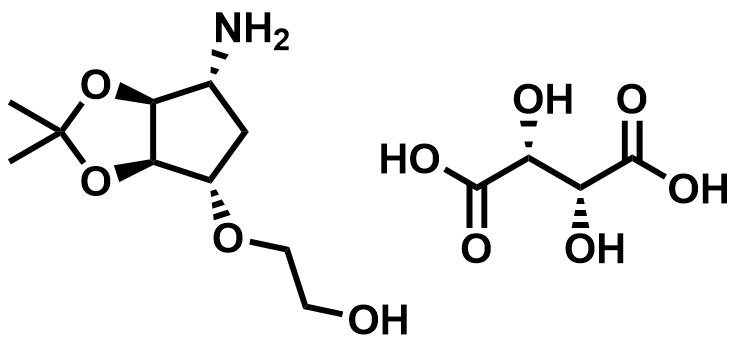 | Intermediate | Commercial |
| 936623-90-4 | Sacubitril/Valsartan | 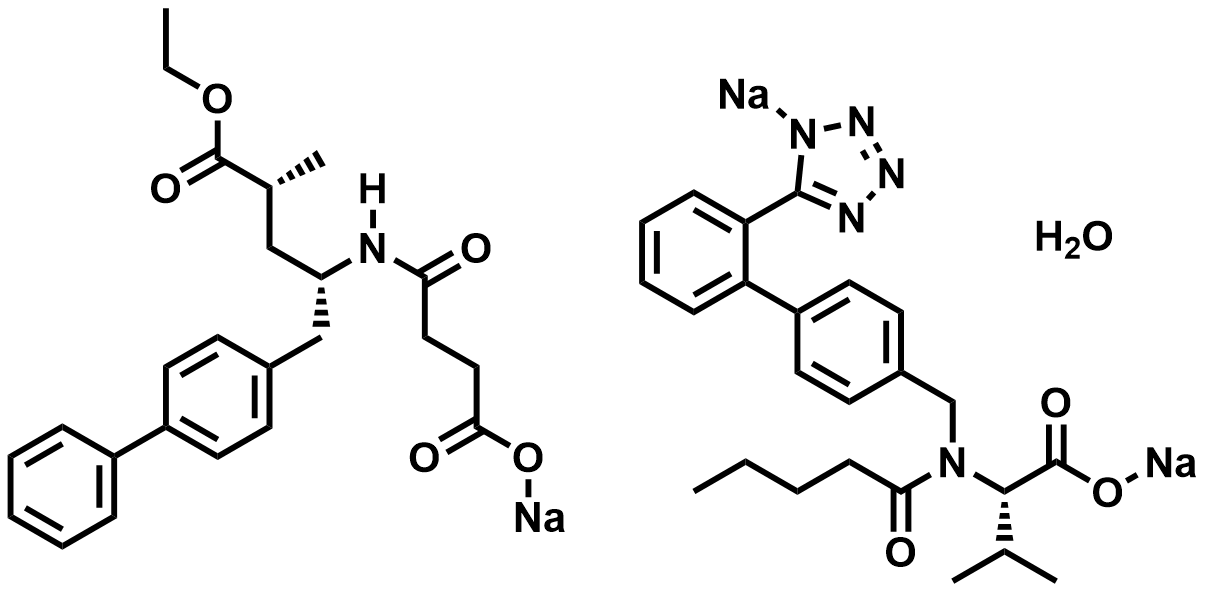 | Heart failure | DMF in Preparation |
| 1426129-50-1 / 1012341-50-2 / 149690-12-0 | Sacubitril Intermediates | 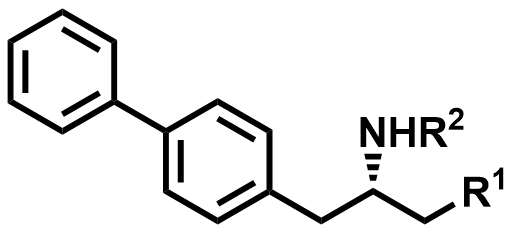 | Intermediates | Commercial |
| 1985606-14-1 | Baloxavir Marboxil | 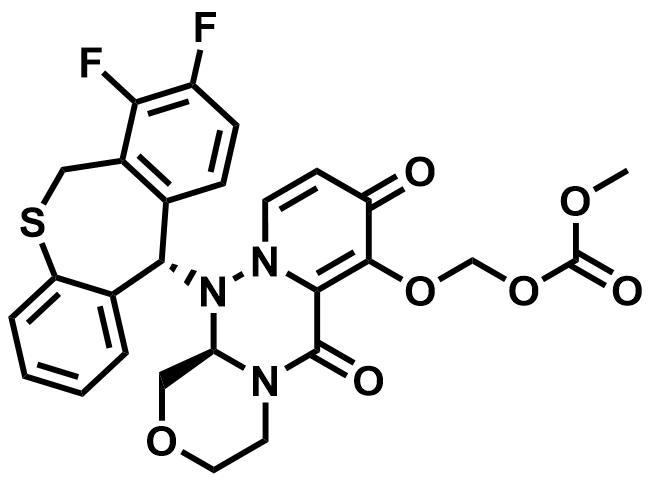 | Influenza | DMF in Preparation |
| 2189684-44-2 | Lenacapavir | 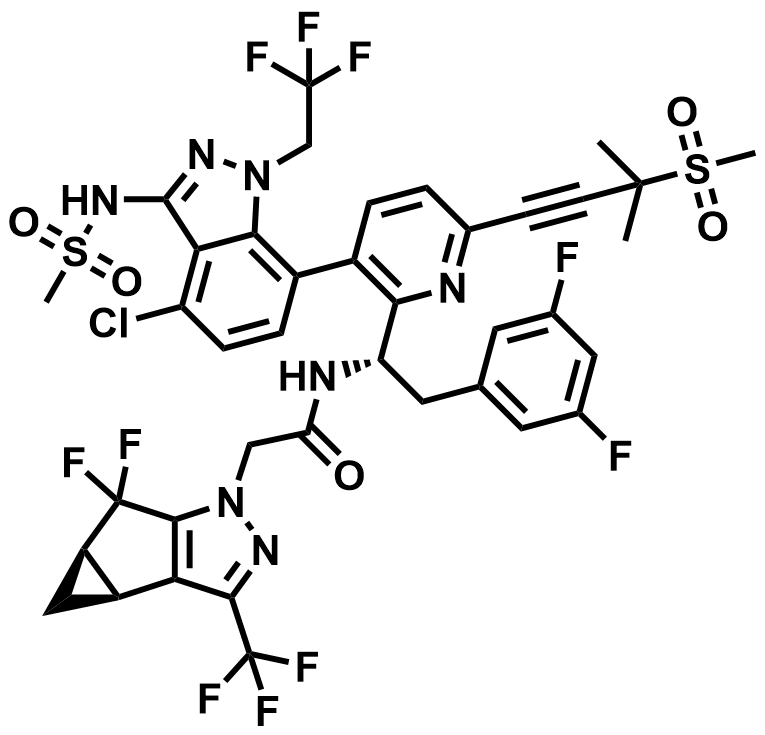 | HIV PrEP / MDR HIV-1 | Pilot |
| 1192500-31-4 | Avibactam | 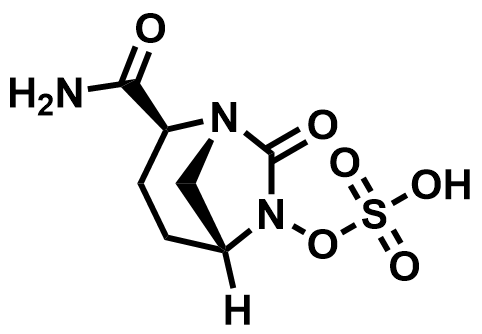 | cIAI / cUTI | Pilot |
| 1416134-48-9 / 63088-78-8 | Avibactam Intermediates |  | Intermediates | Commercial |
| 881681-01-2 | Vonoprazan Fumarate | 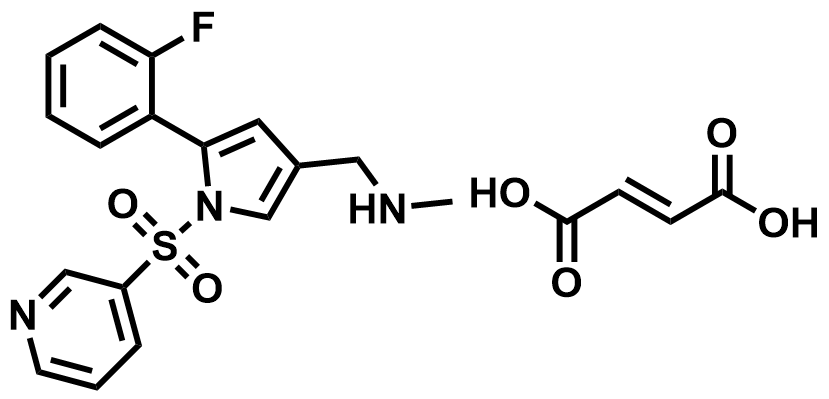 | Reflux esophagitis | DMF in Preparation |
| 942195-55-3 | Tegoprazan | 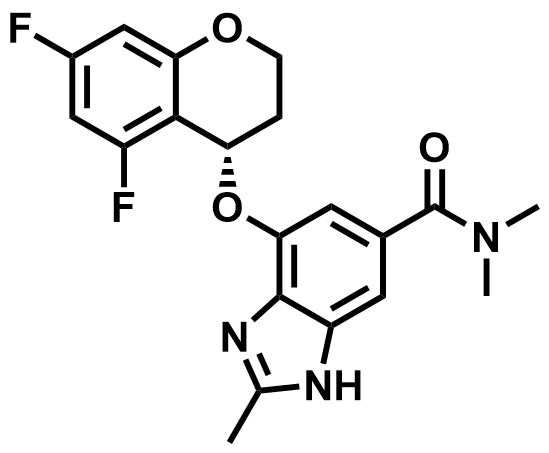 | GERD | DMF in Preparation |
| 864821-90-9 | Eluxadoline | 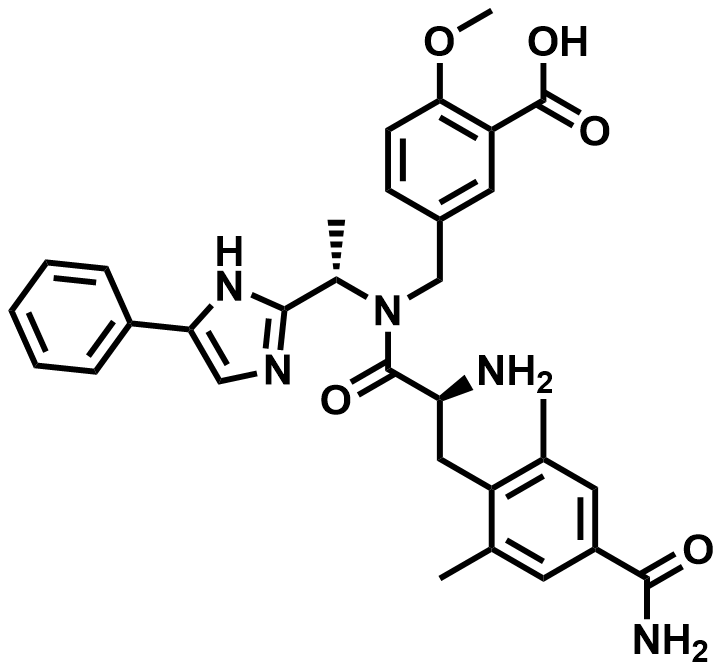 | IBS | Pilot |
| 439087-18-0 | Elobixibat | 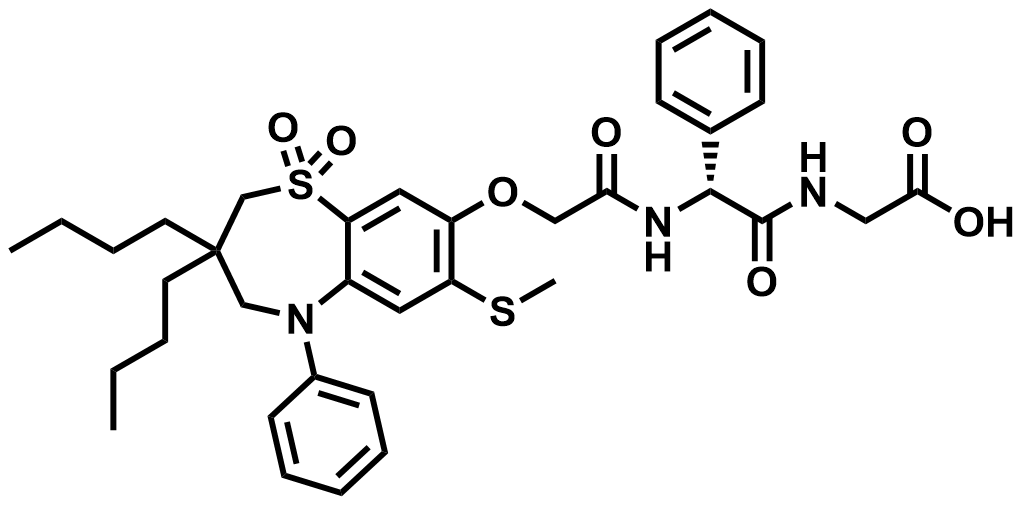 | GI prokinetic | Pilot |
| 1213269-98-7 | Vatiquinone | Friedreich’s Ataxia | Pilot | |
| 709031-45-8 / 361440-67-7 | Saxagliptin Intermediates | 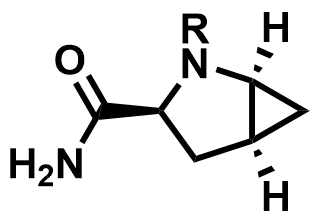 | Type 2 diabetes | Commercial |
| 800412-56-0 | Paxlovid Intermediate | 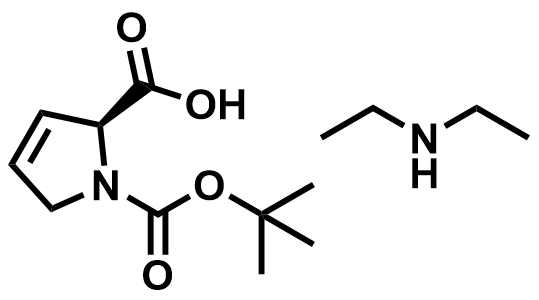 | COVID-19 | Commercial |
| 196597-78-1 | Ramelteon Intermediate | 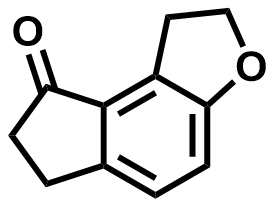 | CNS / Insomnia | Commercial |
| 230615-59-5 / 230615-52-8 | Varenicline Intermediates | 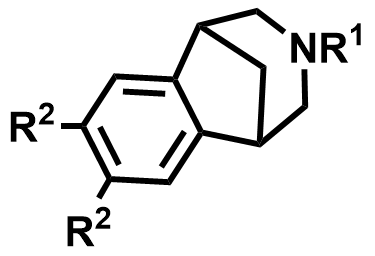 | Smoking cessation | Commercial |
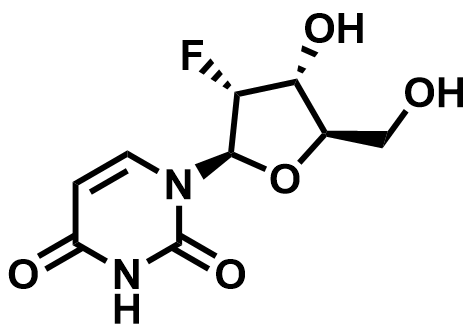
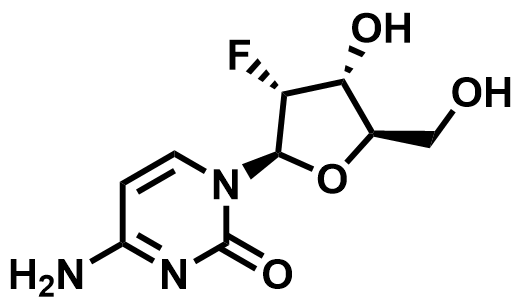
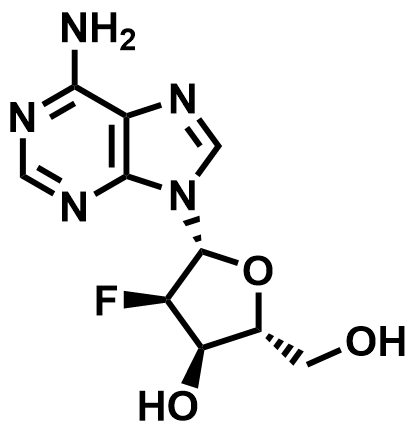
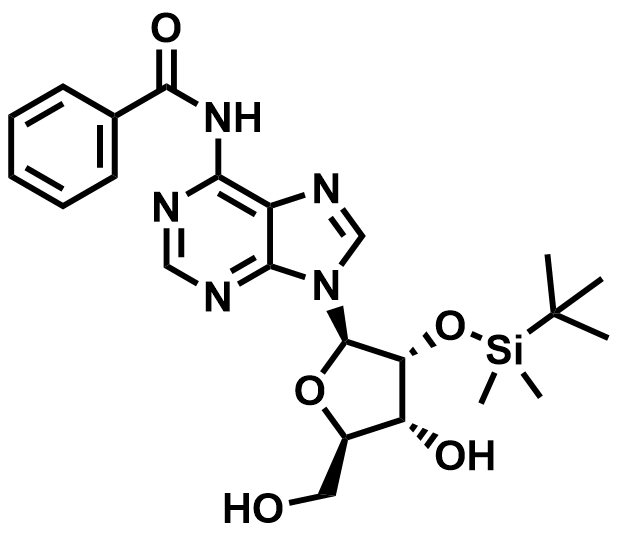
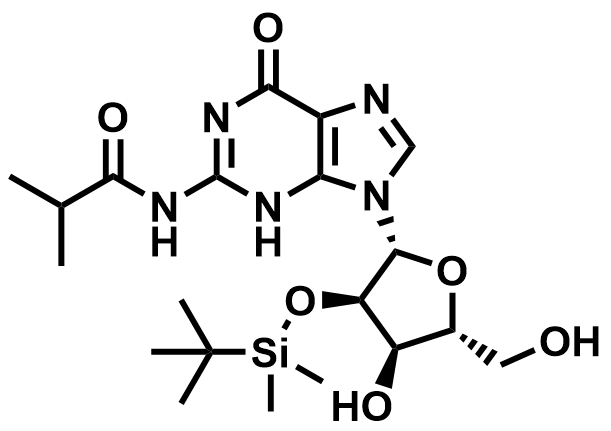
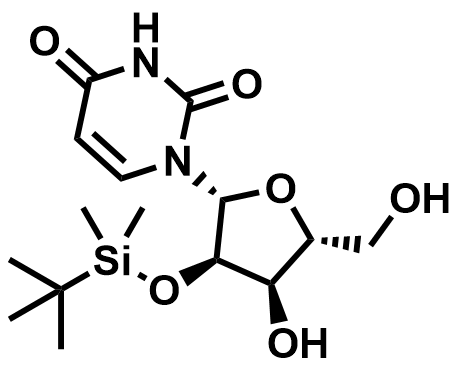
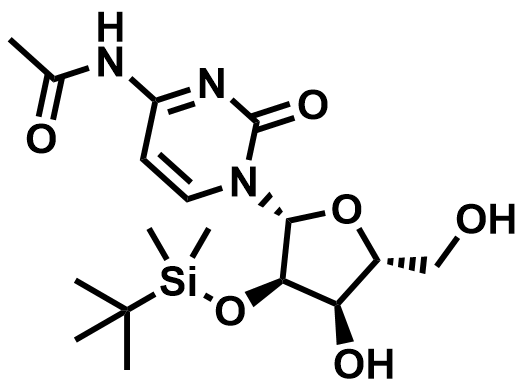
Fluorinated Nucleoside & Protected Nucleoside Intermediates
A growing portfolio of fluorinated nucleosides and TBS-protected nucleoside intermediates for antiviral, oligonucleotide therapeutic, and RNA chemistry applications.
Reaction & Chemistry Capabilities
The sites support a wide range of challenging synthetic transformations:
What OEB / OEL level does the HPAPI facility support?
The dedicated HPAPI facility operates at OEB 5 with an OEL of 0.01 μg/m³. Negative pressure isolators are installed to meet the containment requirements of the most toxic highly potent APIs. OEB 3 and 4 processes are also accommodated.
Which regulatory frameworks does the site comply with?
The site is designed and operated to comply with FDA, EMA, and NMPA GMP standards. A European QP qualification inspection was successfully completed in August 2023. A Japan PMDA on-site inspection is planned for 2026.
What ADC payload-linkers are available with FDA DMF support?
MMAE (MF# 036741), MC-Val-Cit-PAB-MMAE (MF# 036740), and their respective intermediates are all supported with active FDA DMF filings. Exatecan Mesylate (MF# 036708) and Trabectedin (MF# 036724) series are also fully DMF-supported.
What is the total GMP manufacturing capacity?
Site 1 offers over 4,000L of dedicated HPAPI capacity across 5 GMP lines. Site 2 has approximately 480,000L total reactor volume (~175 reactors, 100L–8,000L) for APIs, GMP intermediates and RSMs. Phase II expansion will add a further ~51,000L of HPAPI/ADC payload capacity.
Can the site handle process chemistry at extreme temperatures?
Yes. The facilities support reaction temperatures from −80 °C (using liquid nitrogen cryogenic systems) to +240 °C (using heat transfer oil). This covers the full range required for complex HPAPI synthesis including cryogenic Grignard and asymmetric reactions.
Are Vitamin D analogue APIs and their intermediates available for GMP supply?
Yes. An extensive portfolio of Vitamin D analogue APIs and intermediates is available including calcipotriol, calcitriol, alfacalcidol, paricalcitol, eldecalcitol, maxacalcitol, doxercalciferol, tacalcitol, and calcifediol, with multiple DMF filings including a Japan PMDA DMF for Bazedoxifene Acetate.
Ready to Discuss Your HPAPI or ADC Manufacturing Project?
Our team can support process development, GMP scale-up, regulatory filings and commercial supply. Contact us to discuss your compound, timeline, and containment requirements.